SCHREINER MediPharm is developing customer-specific labels for plastic containers to protect the medicines from contamination.
The growing adoption of plastics as a primary container for medicines in place of glass counterparts is giving rise to concerns about contaminant migration. Soft plastics, in particular, could allow the migration of molecules from solvents and other substances contained in inks, adhesives, and film materials over the years. This could result in eye drops in soft plastic bottles, for example, being contaminated or having reduced effectiveness.
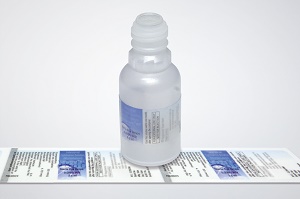 Schreiner MediPharm conducted a study with a renowned, independent test institute to analyze, calculate, and evaluate the migration tendency of various label compositions. The study covered special adhesive systems, materials, and inks. The findings were used to ensure each label design from the company uses harmless materials, allowing it therefore to develop low-migration label solutions for specific applications without additional time and investment costs on the side of the pharmaceutical manufacturer.
Schreiner MediPharm conducted a study with a renowned, independent test institute to analyze, calculate, and evaluate the migration tendency of various label compositions. The study covered special adhesive systems, materials, and inks. The findings were used to ensure each label design from the company uses harmless materials, allowing it therefore to develop low-migration label solutions for specific applications without additional time and investment costs on the side of the pharmaceutical manufacturer.
In addition to the reduced risks for contamination and lower costs, using verifiably harmless low-migration labels raises the probability of a smooth approval from drug administration authorities.
ADIDAS













