Thermoplastic elastomers (TPEs), also known as thermoplastic rubbers, combine the inherent benefits of plastic and rubbery materials. The application of TPEs in the medical field has further been stressed amidst the current coronavirus pandemic with global market for TPEs, assessed at $18.9 billion in 2020, expected to reach $24.2 billion by 2026, rising at a CAGR of 5.2% from 2020-2026, according to a report by Reportlinker.com. In terms of regional market, the United States remains the biggest market at an estimated $5.1 billion in 2021, accounting for 27.09% of the world market for TPEs. China, the world second largest economy, is forecast to reach an estimated size of $4.9 billion in the year 2026, with a CAGR of 7.9% through the analysis period. Among the other noteworthy geographic markets are Japan and Canada, each forecast to grow at 2.8% and 4.7%, respectively, over the analysis period. Within Europe, Germany is forecast to grow at approximately 3.2% CAGR while rest of European market will reach $5.6 billion by the end 2026, according to the report.
The extruded, recyclable, coloured, moulded and eco-friendly attributes of TPEs make them safe for use in applications such as food packaging and medical sector. Rising demand for bio-based TPEs and the growing use of TPEs in medical industry is also poised to favour market growth.
Sustainable materials that can withstand disinfection
In the area of thermoplastics, Avient Corporation has launched three patent-pending grades for the point-of-sale and consumer markets. The new Edgetek™ ET8900 CR series consists of semi-crystalline thermoplastic grades formulations that feature outstanding chemical and crack resistance to common consumer disinfectants versus conventional alternative polymers such as PC/ABS, PC/PET, and PC/PBT.
These materials were developed in response to consumer and industrial market demands for materials that can withstand increased cleaning and disinfecting protocols. Formulations were tested using an adapted ASTM D543 method for environmental stress cracking resistance (ESCR). Results showed that these materials provide better chemical resistance and property retention versus conventional PC blend alternatives after exposure to common consumer disinfectants, including Lysol® all-purpose cleaner, Formula 409® heavy duty degreaser, and Clorox® disinfectant wipes.
“The pandemic has given rise to more frequent cleaning and disinfecting, and this new normal is fuelling demand for materials that can endure increased chemical exposure without degrading,” said Matthew Mitchell, Director, Global Marketing, Specialty Engineered Materials at Avient. “This is especially true for public, high-touch surfaces that are being wiped or sprayed at increasing rates. These new grades offer application and design engineers a durable solution that reduces disinfectant-related failures, like cracking and crazing, so products and surfaces can last longer and look better.”
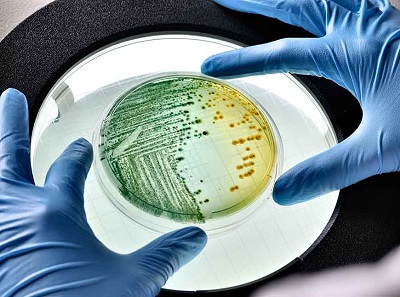
Avient’ GLS TPEs with antimicrobial additives are proven to protect finished parts
from microbial growth on both textured and smooth surfaces.
Avient also unveiled three proprietary, TPE formulations that contain antimicrobial additives to its GLS™ TPE portfolio. GLS TPEs with antimicrobial additives can help extend a product’s useful life, which results in a reduced need to replace it. Additional benefits include preserved surface integrity, increased durability, and minimised odours for a better consumer experience. The GLS TPEs with antimicrobial additives, available as Versaflex™ and OnFlex™ grades, have their potential applications in consumer electronics, personal care item grips, and automotive applications such as cup holder mats and HVAC seals. Tested in accordance with JIS Z2801 and ASTM G21-15 standards, these additives protect moulded plastic parts by inhibiting bacterial growth (99.9% or more) and resisting fungal and mould growth. GLS TPEs with antimicrobial additives are proven to protect finished parts from microbial growth on both textured and smooth surfaces. They are formulated with an EPA-registered antimicrobial from a trusted leader in microbial control solutions, Lonza LLC. This additive chemistry is the same one that has been used for decades in personal care products such as anti-dandruff shampoo.
TPE formulations for medical tubes
Where off-the-shelf tubes used to be in demand simply to keep the development times and costs as low as possible and obtain results fast, preference today is often given to customised, tailor-made solutions, e.g. when it comes to obtaining multi-layer tubes which can be manufactured in a co-extrusion process. An estimated 90% of tube applications today are tailor-made, and that is why instead of manufacturers of basic extruded tubes, there is a reliance today on those which are specialised and offering complex solutions with a high degree of compliance and smooth project management.
The range of materials available for manufacturing medical tubes is enormous: from COC and EVA, HD-PE and LD-PE through PA6.12, PA 11 and 12, PEBA, POM, PVC and PVDF to TEEE, TPU and TPE materials. But not every material is suitable for all areas of application and not every material combination makes sense for the respective characteristics profile required. The formulations of materials used to handle various tasks ensure biocompatibility, resistance to chemicals/media, anti-buckling, a pleasant surface, transparency, as well as sterilisability.
In a market which is so clearly dominated by a demand for customised solutions, Actega has been offering flexibility and creating customised solutions with its large development team of experienced plastics technicians working to develop new, innovative products from the very first requirement profile stated by the customer through to the marketable product.
First of all, any possible raw materials are analysed in detail and compounded to differing material build-ups with various raw materials in the excellently equipped laboratory. These material approaches are then examined in terms of their various properties and those required by the customer. Thanks to an extensive machine park in its own in-house technical centre and the corresponding application technology, test rods and specimens can be manufactured on injection moulding machines once satisfactory material compositions have been found, thereby testing the processing properties of the compounds. This enables new development products to be tested and parameterized in advance, while accelerating and optimising the individual development steps. Then it can be ascertained on site whether the developed materials display good processing properties and meet customer requirements.
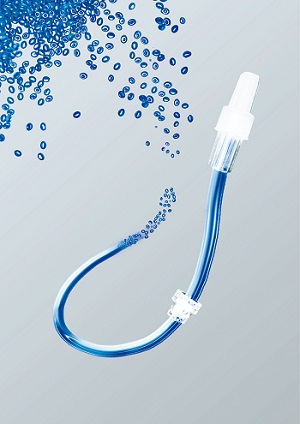
Actega’s ProvaMed® portfolio meet the requirements on medical tubes.
This is how an innovative and steadily growing portfolio has evolved for tube materials. The formulations for Actega’s ProvaMed® portfolio meet the key requirements on medical tubes and can also be tailored to particular customised requirements. Accordingly, these TPE formulations display an excellent degree of transparency. This is a key factor wherever the flow rate of fluids needs to be clearly visible and traceable. This ultra-pure, transparent and elastic material is smooth, flexible and non-buckling. As a result, there is nothing to stop a continuous flow of gases or fluids. Other key factors also include good recovery and attenuation properties enabling tubes to return to their original shape after deformation. The compounds are free of PVC, silicone, latex and plasticisers. This also means that interactions between the tube material and pharmaceutical or endogenous substances which could be harmful to the human organism are in fact avoided. The ProvaMed® formulations for tube materials can be sterilised and display outstanding processing characteristics.
Toughness and chemical resistance
Two leading plastics suppliers to the medical device industry have demonstrated a strong, chemical-resistant bond between TPEs and a clear copolyester substrate known for its toughness and chemical resistance. In 90-degree peel tests of test specimens produced in a two-shot moulding process, three Medalist® medical-grade TPEs from Teknor Apex Company provided durable bonds to four of Eastman’s Tritan™ copolyesters. Specifically formulated to adhere to copolyester and other engineering thermoplastics, the Medalist compounds exhibited high levels of peel strength. The peel tests that showed high peel strengths with adhesive failures demonstrate excellent Tritan TPE over-moulding performance. The combination of high peel strength and adhesive failure is considered desirable for over-moulded applications.
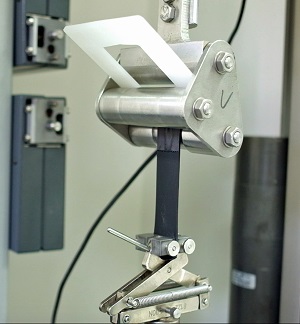
Medalist® medical-grade TPEs from Teknor Apex provided
durable bonds to four of Eastman’s Tritan™ copolyesters.
The bond strength between Medalist TPEs and Tritan exhibited very little loss even after soaking test samples for 48 hours in disinfecting chemicals, including a 70% isopropyl alcohol, Clorox® bleach, and a 20% intralipids solution, which is a fat emulsion used as an intravenously administered nutrient. Medalist compounds tested included three grades: MD-34940, MD-34950, and MD-34959, with Shore A durometers of 42, 50, and 59, respectively. Tritan grades MX711, MX811, MXF321, and MXF421, provided a good range of physical properties. All three Medalist compounds in combination with all four Tritan resins showed very little loss of bond strength after chemical exposure. Over-moulding TPE onto copolyester offers medical device designers a combination of hard and soft materials that are advantageous in medical components. Soft TPEs are desirable for grips, gaskets, seals, and soft-touch elements on rigid medical housings and other components.
“By combining Medalist TPEs and Tritan copolyesters, manufacturers can improve the ergonomics, aesthetics, or functionality of medical devices,” said Dr. Yubiao Liu, global medical technical platform lead, Eastman. “The combination of soft TPEs with rigid copolyester substrates can enhance grip, add texture or comfort, yield a tight seal, or provide damping characteristics.”
Medalist TPEs combine the performance of thermoset rubber with the processing ease of thermoplastics, making them superior alternatives to silicones, PVC, and TPUs. As note by Ross van Royen, senior market manager for Teknor Apex: “We have developed Medalist TPEs to meet the most stringent requirements of the health care industry and produce them in multiple ISO-13485-certified facilities with only FDA-listed food-grade ingredients. All grades are biocompatible in accordance with ISO-10993-5, are RoHS and REACH SVHC compliant, and contain no phthalates, latex proteins, or BPA.”

 iConnectHub
iConnectHub
 Login/Register
Login/Register Supplier Login
Supplier Login


























